You never realize how dependent you are to the grid until the lights go out for a long period of time. Although many of us have batteries and flashlights safely stored for a rainy day, there may be a time during an extended emergency where you run out of your battery stash and have to improvise.
Creating a Voltaic Pile
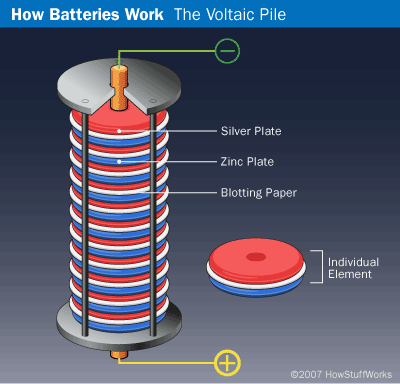
Batteries were previously known as voltaic piles. When Alessandro Volta discovered the first electric battery in 1800, he made a giant stack of alternating layers of zinc, blotting paper soaked in salt water, and silver to create a charge. This wet cell battery creates energy because the redox reaction between the copper and zinc metals allow electrons to flow and each cell provides around 0.6 volts of electric potential.
You can make a simple battery similar to Volta’s using the humble penny. Coins can be converted into makeshift batteries that can drive up to 0.8 volts. Since the battery is a wet cell, the effectiveness will be reduced when the electrolyte evaporates. That said, this can produce more than enough current to provide power to small current devices and many of these penny batteries can be stacked together to produce higher voltages. Here’s what you [amazon asin=B00006IBNJ&template=*lrc ad (right)]need to get started:
(Makes 1 volt)
- 6 pennies – pennies newer than 1982 are 98% zinc
- sand paper or orbital sander
- adhesive remover such as Goo-Gone
- Cardboard pieces
- Vinegar
- Electrical tape
- Zinc washers
LEGALITIES: Some people have asked about the legality of treating pennies in this manner. The federal law states that there are exceptions made for use as “educational, amusement, novelty, jewelry, and similar purposes as long as the volumes treated and the nature of the treatment make it clear that such treatment is not intended as a means by which to profit solely from the value of the metal content of the coins.”
Reprinted from Ready Nutrition.
[amazon asin=B001CD9RLW&template=*lrc ad (left)]
[amazon asin=B006J36V8M&template=*lrc ad (left)]
[amazon asin=B000RX0W3K&template=*lrc ad (left)]
[amazon asin=B005S28ZES&template=*lrc ad (left)]




